The urgent need for African research collaboration on medicine quality
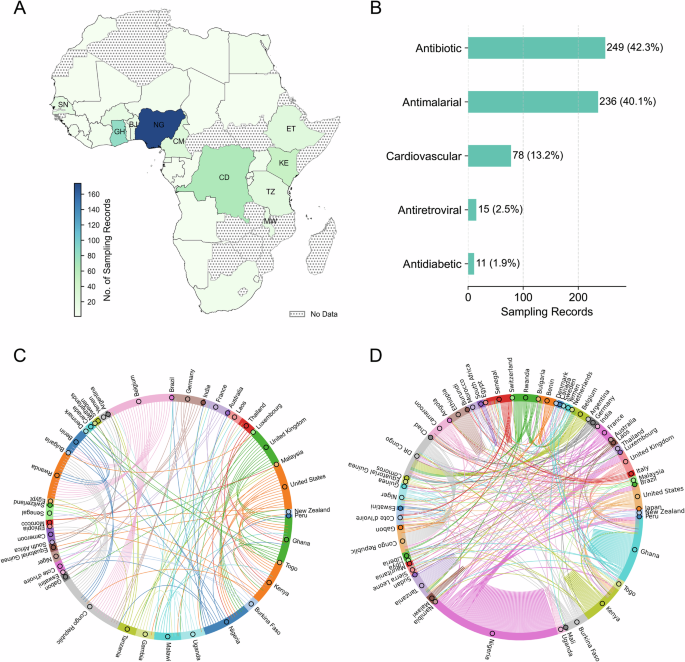
To grasp the extent of this evidence gap, we analysed 268 original research articles on SF antibiotics, antidiabetics, antimalarials, antiretrovirals, and cardiovascular medicines worldwide, published between 1963 and 2021 (as the dataset only goes up to this year at the time of access), using data from the Medicine Quality Scientific Literature Surveyor8. Just over 50 percent of the articles included African sampling records, and those records disproportionately focused on a few countries: Nigeria, Ghana, the Democratic Republic of Congo, and Kenya– with 19 out of 54 African countries lacking sampling records (Fig. 1). Most African countries are absent from the literature.
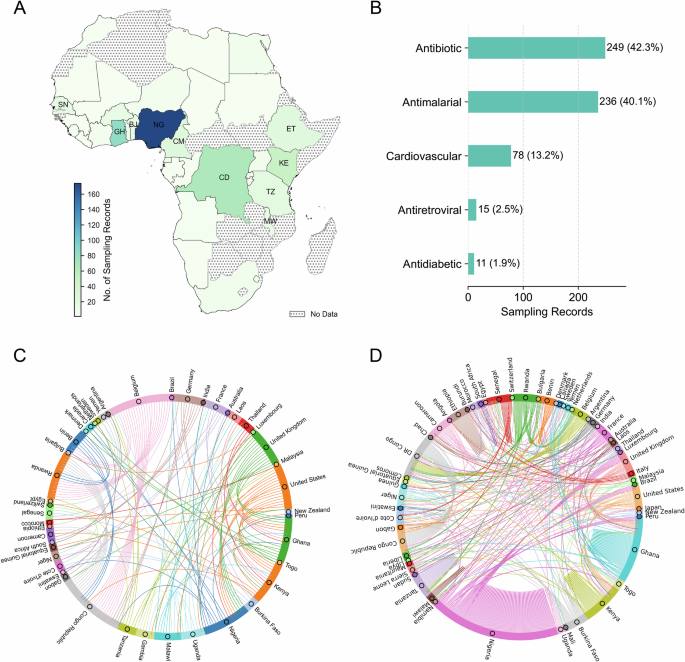
The Surveyor is an online database developed by the Infectious Diseases Data Observatory (IDDO) and the Medicine Quality Research Group (MQRG)8, which aggregates and curates data from various sources, categorized by publication type as original research article, report, lay press, etc. We first identified 268 original research articles studying SF (either substandard or falsified or both) ‘Antibiotic’, Antidiabetic’, ‘Antimalarial’, ‘Antiretroviral’, and ‘Cardiovascular’ medicines worldwide between 1963 and 2021. A Out of these 268 original research articles, 150 articles consist of 589 sampling records of SF medicines in Africa. The distribution of these 589 sampling records on SF medicines is uneven, with the top ten countries being Nigeria (NG, N = 174, sampling ratio\(\approx\)30%), Ghana (GH, N = 83, sampling ratio\(\approx\)14%), Democratic Republic of the Congo (CD, N = 66, sampling ratio\(\approx\)11%), Kenya (KE, N = 49, sampling ratio\(\approx\)8%), Cameroon (CM, N = 26, sampling ratio\(\approx\)4%), Ethiopia (ET, N = 23, sampling ratio\(\approx\)4%), Tanzania (TZ, N = 20, sampling ratio\(\approx\)3%), Senegal (SN, N = 17, sampling ratio\(\approx\)3%), Benin (BJ, N = 15, sampling ratio\(\approx\)3%), and Malawi (MW, N = 14, sampling ratio\(\approx\)2%). B The counts of the five sampled categories of SF medicines in Africa show that most SF medicines sampling records within the 589 records are related to antibiotics and antimalarials. C Based on the 268 selected articles studying SF medicines worldwide, we constructed a collaboration network. This chord diagram indicates collaboration between countries whenever the authors’ institutions from different countries appear in the same article. Each node represents a country, with its size proportional to the number of unweighted edges connected to it (D). Notably, collaborations in the study of SF medicines were primarily composed of researchers in the Global North, working with researchers in just 23 African countries. While there were a few connections with countries in Asia and Latin America, intra-Africa collaboration on this issue was scarce, accounting for only 33 out of 141 edges (23.4%). D The selected 150 original research articles sampling SF medicines in Africa consisted of 185 unique sampling records containing information on authors’ countries and sampled African countries. This was used to construct an investigation network wherein the links indicate investigation directions originating from researchers’ countries to the sampled African countries. The per-country table for (A) and edge lists for (C, D), and the data collection procedure from the Surveyor can be found in the Supplementary Data and Code.
Moreover, as shown in Fig. 1, the types of medicines studied in Africa are narrowly clustered around antibiotics and antimalarials. Other drug classes, including those targeting non-communicable diseases, are significantly underrepresented. This uneven evidence base undermines the capacity of African regulators to make informed decisions. It hampers efforts to coordinate surveillance, target interventions, and evaluate the effectiveness of reforms.
Many investigations were conducted locally within the researchers’ home countries, as shown in Fig. 1D by the self-connected edges (128 out of 381 edges, 33.6%), predominantly in a few African countries. Notably, there were many investigations led by countries in the Global North. The figure reveals that research on SF medicines in Africa focused on a few African countries and was dominated by forces outside the continent, with the Global North playing the leading role, while intra-Africa collaboration remained underdeveloped.
Meanwhile, given the lack of data on post-2021 publications, the patterns of country and drug-class coverage in Fig. 1 might differ slightly, particularly due to COVID-19 pandemic impacts. Additionally, the dataset was primarily funded by Western funders and includes mainly English-language literature, potentially introducing bias by missing non-English and local language publications. This limitation highlights another inequality in public health database infrastructure in Africa.
link